Sanitary Mechanical Seal

Is a Sanitary Mechanical Seal suitable for Aseptic Processes?
An aseptic process is one that requires a sterile environment and a sanitary mechanical seal installation could well be a part of it.
For a mechanical seal to become sanitary the seal must be cleaned in a way
that assures that harmful organisms have been purged from all surfaces, cracks, pools, and reservoirs that naturally exist in mechanical seals. Some of the methods used to produce a sanitary condition in a mechanical seal can cause damage to the sealing surfaces or the materials from which the seal is made. The sanitary mechanical seal designer must take care to understand the cleaning method to be used and thus design the seal configuration and materials to tolerate those conditions.
There are 2 aseptic processes; Biotechnology Processing and Fill Finish.
In the case of Biotechnology processing, it specifically processes living organisms. These living organisms are grown in tanks and vessels where they are fed nutrients and oxygen at an optimum rate for growth and
reproduction. The vessel conditions during processing are not demanding and well within the capabilities of some lower technology seal. However, the actual seal design is critical that it takes into account CIP and SIP between batches without having to remove the actual seal and maintain the suitably low bio-burden condition of the equipment.
A properly designed seal will increase the repeatability of CIP and SIP while minimizing downtime of the equipment. This is even more important for the bottom entry mixer configuration which tends to be a
standard in the bioprocessing industry.
In the case of a Fill Finish, which entails making up the end product for distribution e.g pill form, injectables or other dosage method etc
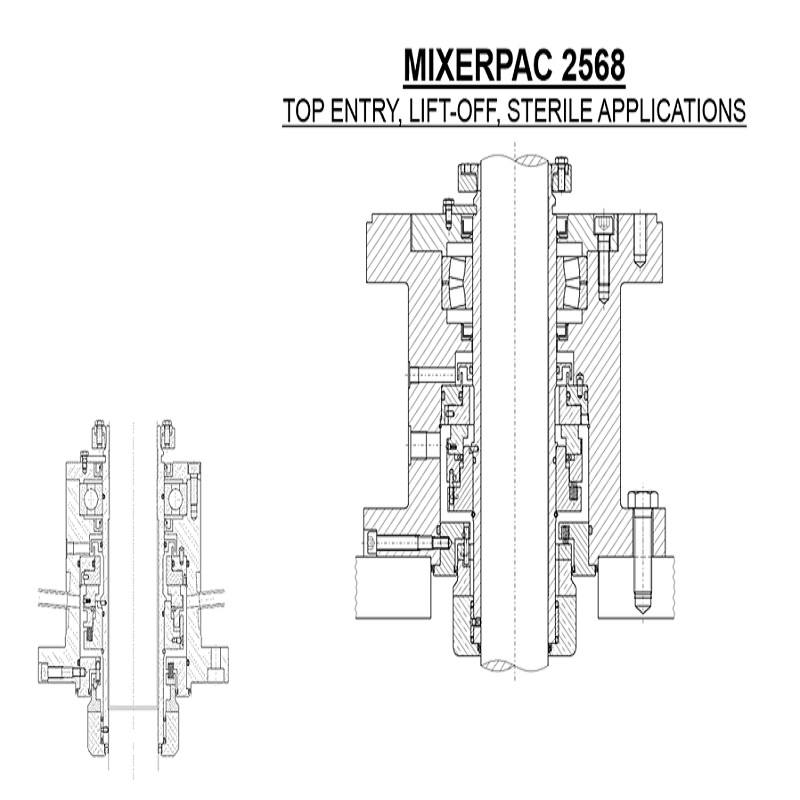
Fill Finish typical application is a mixer pac 2568 (gas lift off seal) as processes wont tolerate wear particulate. Mechanical Seal faces are located in the vessel for better cleaning. As oppose to bottom entry mechanical seal faces which are in the actual process.
Demands for aspectic process, sanitary mechanical seals & reliability;
- low / zero wear
- drainability
- sterilization
- Process Isolation
What are Bio-Reactor Seals?
Bio-reactor seals are a type of mechanical seal installed on a Bio-Reactor Mixer. There are single mechanical seals and double mechanical seals, the latter is where the barrier fluid is clean steam condensate (WFI).
Bottom entry mixers / agitators are more common in industry.
How we can help you?
Flexachem provides a full range of Flowserve Sanitary Mechanical Seals; single, dual, dry contacting, dry non-contacting, and liquid lubricated seals.
We supply sanitary mechanical seals into the pharmaceutical, bio-technology, semi-conductor and general industries.
Depending on your particular application, if you are looking mechanical seals to suit a pump application; Mechanical Seal for Pumps, or if you are trying to source mechanical seals for an agitator or tank mixer; Mechanical Seal for Agitator.
We also provide Sealing Supporting Systems – reservoirs, barrier systems, piston transmitters, gas panels, seal coolers, circulators and fill carts.
Our Mechanical Seal specialists can advise you on the appropriate selection of a seal support system which will deliver years of reliable service and operating cost savings in the longer term.
Contact our Mechanical Seal Team:
Chris Sheeran (External) – mob: 087 755 9936
Internal Team
Rory Cremin – Tel: 021 461 7213
Tel: 021 461 7200